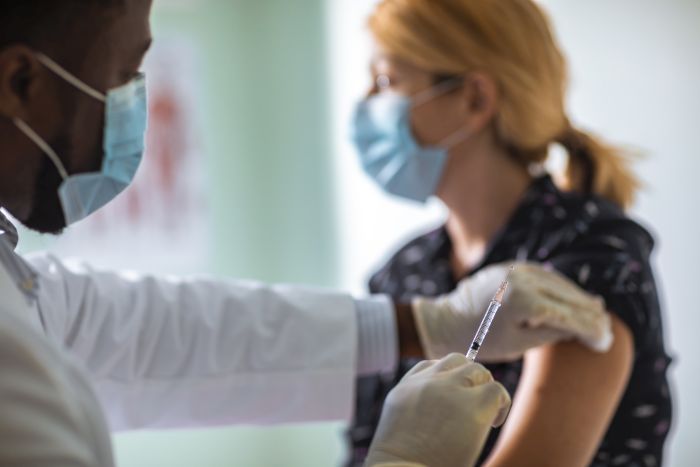
The world has learned a lesson or two over the past year about the vulnerability of supply chains. Anyone who has experienced a delay in the delivery of an urgently needed product – and that’s most of us – understands the potential impact to people and society.
A Supply Chain Challenge of New Proportions
The pharmaceuticals industry is no different. Numerous pharma research and development companies have created and tested vaccines at record speed. Now they are facing the next challenge. To get the pandemic under control, about 7.6 billion earthlings need a shot – or two – in the arm that can come none too soon as the virus mutates and becomes more and more virulent. The demand is without precedent, stretching the supply chains of the companies that produce COVID-19 vaccines to their limits.
Supply chains in the pharmaceutical industry are complex and haven’t always been the most robust, according to Bruce Y. Lee, professor of Health Policy and Management at the City University of New York Graduate School of Public Health and Health Policy. The reason, according to Lee, was often inadequate storage, transportation, and personnel capacity, leading to major bottlenecks and the discarding of many vaccine doses.
But the world’s vaccine producers are not starting from zero. Data-heavy industries like pharmaceuticals have matured, and many of the global players have digitalized their production to achieve production scale and flexibility.
Major Vaccine Producers Overwhelmingly Run SAP
Today, 18 of the world’s 20 major vaccine producers are already running their production on SAP solutions that cover the end-to-end process from manufacturing to controlled distribution to administration and post-vaccine monitoring. One of those producers is German vaccine manufacturer CureVac, whose COVID-19 vaccine is currently in the testing phase. “We need SAP to manufacture and distribute our vaccine on a global scale,” says Pierre Kemula, CFO at CureVac.

Video by Tilman Goettke and Klaus Boeckle, SAP
Another producer, Moderna, which began shipping its COVID-19 vaccine in January, is relying on SAP to help distribute hundreds of millions of vaccine doses. The company is running digital supply chain solutions from SAP to help with serialization and distribution. SAP Advanced Track and Trace for Pharmaceuticals is helping Moderna to comply with international legislation intended to prevent counterfeit drugs from reaching patients. The application provides a corporate serialization repository, serial number management, and regulatory reporting capabilities. To work with its supply chain partners, namely contract manufacturers and wholesalers like AmerisourceBergen, Moderna leverages SAP Information Collaboration Hub for Life Sciences, a public cloud network that enables collaboration for pharmaceutical supply chain trading partners. In the U.S. alone, the hub is already processing more than 70 percent of drug volume for verification.
Similar tools are in use at AstraZeneca, Sanofi, GlaxoSmithKline (GSK), and Pfizer, all pharmaceutical companies working on COVID-19 vaccines. Here are just a few examples:
- Over 90 percent of global revenue from GSK’s pharmaceutical and vaccine businesses is managed with SAP software, enabling the company to leverage the data it collects and develop insights from that information to improve operations. GSK also uses SAP Ariba to help procure the raw materials that make up its vaccine formulas.
- AstraZeneca uses SAP Work Manager to oversee its manufacturing equipment and help managers predict when machines require maintenance. This has resulted in a six to eight percent increase in worker efficiency, according to Michael Zboray, lead business analyst at AstraZeneca.
- Sanofi, a French drug manufacturer, introduced SAP S/4HANA in 2017 to help standardize and harmonize processes, support the development of standard solutions, and improve efficiency.
Vaccines Require a Supply Chain with Speed, Transparency, and Flexibility
Once a producer has finalized its immunization formula, reliable production and logistics are the next hurdle. But the distribution and administration of vaccines at the scale of COVID-19 is a monumental challenge. There is much at stake because disruptions in the supply chain can prevent the delivery of the vaccine and negatively impact public trust. Delays are paid for with human lives and push out the prospect of herd immunity. Reacting quickly to unexpected interruptions is key and is only possible if the entire supply chain, from procurement to production and delivery, is managed end-to-end.
According to Dr. Oliver Nürnberg, chief product owner of SAP for Life Sciences at SAP, three characteristics differentiate the COVID-19 supply chain from a typical pharmaceutical supply chain and make tight coordination a must.
First, the temperature of most vaccines needs to be closely controlled, generally between 2° and 8° Celsius or frozen at temperatures within the range of a standard freezer. The temperature of the BioNTech/Pfizer vaccine, however, must be held at minus 70° Celsius, which requires special handling along its entire distribution route to the vaccination centers. “Real-time temperature monitoring is a huge asset here and allows the producer to react quickly if any vaccine has spoiled during shipping,” says Nürnberg.
Second, because the demand for the vaccines will be greater than the supply for quite some time, the supply chain for COVID-19 operates in “push” rather than “pull” mode. Governments are taking as much vaccine as they can get their hands on and no standard ordering is required.
Third, the players and conditions are different. Recipients of COVID-19 vaccine shipments are generally government authorities and not the pharmacies that routinely handle delicate pharmaceuticals. Medical personnel, equipment, patients, and vaccines must come together at newly installed vaccination centers just in time, under the right conditions, and in the right quantities.
All of these characteristics place demands on vaccine management teams to ensure speed, transparency, and flexibility.
SAP’s Vaccine Collaboration Hub Supports German Vaccination Campaign
To support large-scale vaccination programs like the world is experiencing with COVID-19, SAP recently launched its vaccine collaboration hub (VCH), an extension to its business network. It covers the end-to-end process from manufacturing to controlled distribution to administration as well as post-vaccine monitoring. Through cloud-based applications from SAP, such as SAP Logistics Business Network, SAP Integrated Business Planning for Supply Chain, SAP S/4HANA, SAP Analytics Cloud, and cloud solutions from SAP, the VCH provides transparent insight into the vaccination process.
“Humanitarian logistics is a big challenge in our fight against the pandemic,” says Lars Werthmann from the German Red Cross (DRK) in Saxony. “SAP’s solutions enable digitized planning, procurement, inventory management, and delivery of the vaccines.” The platform shows availability, supply quantities, and progress, making the entire logistics and distribution process plannable in real-time to support the DRK in vaccinating citizens seven days a week.
“The vaccine collaboration hub allows people to focus on their tasks along the supply chain, whether it’s planning, ordering, producing, storing, transporting, delivering, tracking, inspecting, or injecting the vaccines,” says Katrin Lehmann, head of Customer Innovation and Maintenance at SAP. “The ultimate goal is to save lives, restart the economy, and bring people back together again.”
Sarah Heinz, Klaus Boeckle, and Tilman Goettke contributed to this story.
This article first appeared on the SAP Global News Center.